A complex unit for a complex disease: the HCM-Family Unit
Accepted: November 30, 2021
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
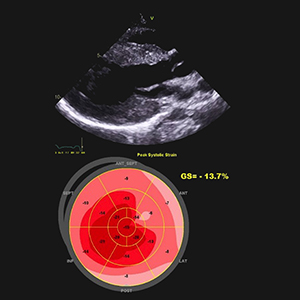
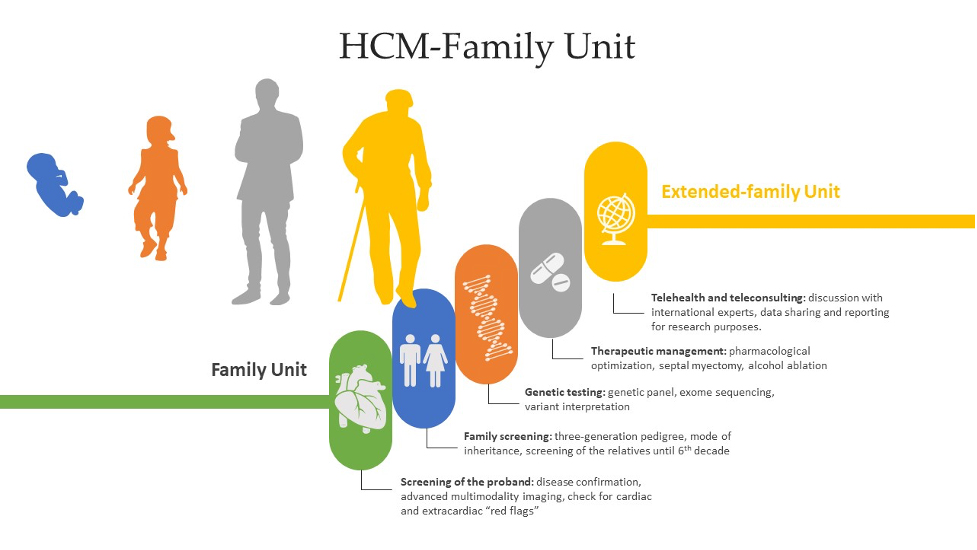
Hypertrophic cardiomyopathy (HCM) is a group of heterogeneous disorders that are most commonly passed on in a heritable manner. It is a relatively rare disease around the globe, but due to increased rates of consanguinity within the Kingdom of Saudi Arabia, we speculate a high incidence of undiagnosed cases. The aim of this paper is to elucidate a systematic approach in dealing with HCM patients and since HCM has variable presentation, we have summarized differentials for diagnosis and how different subtypes and genes can have an impact on the clinical picture, management and prognosis. Moreover, we propose a referral multi-disciplinary team HCM-Family Unit in Saudi Arabia and an integrated role in a network between King Faisal Hospital and Inherited and Rare Cardiovascular Disease Unit-Monaldi Hospital, Italy (among the 24 excellence centers of the European Reference Network (ERN) GUARD-Heart).
Graphical Abstract
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.
Similar Articles
- Mario Tamburrini, Unnati Desai, Giovanni Fanti, Angelo Scarda, Francesca Zampieri, Umberto Zuccon, Parikshit Thakare, Marco Contoli, Alberto Papi, Medical thoracoscopy without pleural fluid: How I do it , Monaldi Archives for Chest Disease: Vol. 92 No. 1 (2022)
- Gennaro Liccardi, Luigino Calzetta, Manlio Milanese, Maria Beatrice Bilò, Maria Vittoria Liccardi, Maria Gabriella Matera, Paola Rogliani, Anxiety and depression in adolescents with asthma and in their parents. Is an increased basal cholinergic tone a possible further reason to explain the negative impact on asthma control? , Monaldi Archives for Chest Disease: Vol. 90 No. 1 (2020)
- William Newmarch, Angelica Puopolo, Madina Weiler, Brian Casserly, Hamman-Rich syndrome: a forgotten entity , Monaldi Archives for Chest Disease: Vol. 87 No. 1 (2017)
- Floriana Caccamo, Simone Saltini, Enrico Carella, Roberto Carlon, Cristina Marogna, Vito Sava, The measure of effectiveness of a short-term 2-week intensive Cardiac Rehabilitation program in decreasing levels of anxiety and depression , Monaldi Archives for Chest Disease: Vol. 88 No. 1 (2018)
- Martina Maria Seminara, Dina Visca, Alice Claudia Repossi, Antonio Spanevello, Mycobacterium chimaera: a case report from Italy , Monaldi Archives for Chest Disease: Early Access
- Federico Piffer, Guido Levi, Giampietro Marchetti, Chiara Barbieri, Immune reconstitution inflammatory syndrome in tuberculous pleurisy and ulcerative colitis: a case report , Monaldi Archives for Chest Disease: Vol. 89 No. 3 (2019)
- Eleni D. Eleftheriadou, Maria Saroglou, Nikolaos Syrigos, Ellias Kotteas, Marousa Kouvela, The role of immunotherapy in patients with lung cancer and brain metastases: a narrative review of the literature , Monaldi Archives for Chest Disease: Early Access
- Danish Abdul Aziz, Muhammad Aqib Sajjad, Haissan Iftikhar, Clinical outcomes of children with acute asthma managed with intravenous magnesium sulphate outside intensive care setting , Monaldi Archives for Chest Disease: Early Access
- Matteo Gelardi, Rossana Giancaspro, Francesca Fortunato, Michele Cassano, Italian survey on the effectiveness of halotherapy administered via the Aerosal® system , Monaldi Archives for Chest Disease: Early Access
- Fabiola B. Sozzi, Laura Iacuzio, Marta Belmonte, Marco Schiavone, Francesca Bursi, Elisa Gherbesi, Frank Levy, Ciro Canetta, Stefano Carugo, Early diagnosis of cardiomyopathies by cardiac magnetic resonance. Overview of the main criteria , Monaldi Archives for Chest Disease: Vol. 92 No. 4 (2022)
<< < 20 21 22 23 24 25 26 27 28 29 > >>
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/monaldi.2021.2147
https://doi.org/10.4081/monaldi.2021.2147




