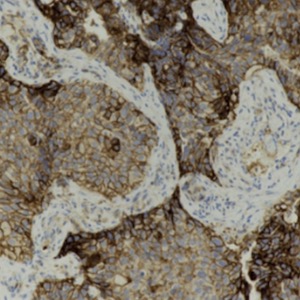
Complete response to pembrolizumab as a single agent in a patient with stage III NSCLC with high PD-L1 expression: a case report
Submitted: September 27, 2022
Accepted: November 14, 2022
Published: November 25, 2022
Accepted: November 14, 2022
Abstract Views: 1184
PDF: 301
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Similar Articles
- Saulius Diktanas, Oleksandr Korotych, Yuliia Sereda, Ogtay Gozalov, Olga Rubcova, Jay Achar, Factors associated with time to sputum culture conversion of rifampicin-resistant tuberculosis patients in Klaipeda, Lithuania in 2016-2019: a cohort study , Monaldi Archives for Chest Disease: Vol. 91 No. 1 (2021)
- Federico Raimondi, Caterina Conti, Luca Novelli, Francesco Tarantini, Giuseppe Ciaravino, Piermario Scuri, Aurelia Grosu, Daniela Chinaglia, Lorenzo S.C. Grazioli, Andrea Gianatti, Ferdinando L. Lorini, Michele Senni, Fabiano Di Marco, A 57-year-old man with rapidly progressive pulmonary hypertension , Monaldi Archives for Chest Disease: Vol. 92 No. 1 (2022)
- Avneet Garg, Rakendra Singh, Hariharan Iyer, Mansimranjit Kaur, Surbhi Surbhi, Ashish Jindal, Saranpal Singh, Avtar Singh Bansal, Hem C. Sati, Vinita Jindal, To study heterogeneity in SARS-CoV-2 IgG response titre in patients recovered from COVID-19 , Monaldi Archives for Chest Disease: Vol. 92 No. 2 (2022)
- Zehra Naseem, Muhammad Mussab Khakwani, Maaha Ayub, Ahmed Ayaz, Bushra Jamil, Ainan Arshad, SARS-CoV-2 induced coagulopathy and potential role of anticoagulation: Scoping review of literature , Monaldi Archives for Chest Disease: Vol. 92 No. 4 (2022)
- Michele Vitacca, Beatrice Salvi, Marta Lazzeri, Elisabetta Zampogna, Giancarlo Piaggi, Piero Ceriana, Serena Cirio, Luigino Rizzello, Grazia Lacala, Angelo Longoni, Vittoria Galimberti, Patrizia D'Ambrosio, Enrica Pavesi, Giuseppe La Piana, Antonella Sanniti, Alessandro Morandi, Manoel Vallet, Mara Paneroni, Respiratory rehabilitation for patients with COVID-19 infection and chronic respiratory failure: a real-life retrospective study by a Lombard network , Monaldi Archives for Chest Disease: Vol. 92 No. 3 (2022)
- Manu Dogra, Surabhi Jaggi, Deepak Aggarwal, Seema Gupta, Varinder Saini, Jasbinder Kaur, Role of interleukin-6 and insulin resistance as screening markers for metabolic syndrome in patients of chronic obstructive pulmonary disease. A hospital-based cross-sectional study , Monaldi Archives for Chest Disease: Vol. 92 No. 3 (2022)
- Nikhilesh Ladha, Pankaj Bhardwaj, Nishant Kumar Chauhan, Kikkeri Hanumantha Setty Naveen, Vijaya Lakshmi Nag, Dandabathula Giribabu, Determinants, risk factors and spatial analysis of multi-drug resistant pulmonary tuberculosis in Jodhpur, India , Monaldi Archives for Chest Disease: Vol. 92 No. 4 (2022)
- Sara Parini, Paolo Spina, Esther Papalia, Renzo Boldorini, Michele Abruzzese, Ottavio Rena, Primary seminoma arising in the posterior mediastinum: a diagnostic challenge , Monaldi Archives for Chest Disease: Vol. 92 No. 2 (2022)
- Ravindra Nath, Neeraj Kumar Gupta, Amandeep Jaswal, Sparsh Gupta, Navjot Kaur, Santvana Kohli, Anirudh saxena, Pranav Ish, Rohit Kumar, Poornima Tiwari, Mukesh Kumar, Jugal Kishore, Geeta Yadav, Fellisha Marwein, Nitesh Gupta, Mortality among adult hospitalized patients during the first wave and second wave of COVID-19 pandemic at a tertiary care center in India , Monaldi Archives for Chest Disease: Vol. 92 No. 2 (2022)
- Vijayarangam Narenchandra, Govindaraj Vishnukanth, Dharm Prakash Dwivedi, Munuswamy Hemachandren, Subathra Adithian, Saka Vinod Kumar, Manju Rajangam, Madhusmita Mohanty Mohapatra, Pratap Upadhya, Comparison of efficacy of autologous blood patch pleurodesis versus doxycycline pleurodesis in the management of persistent air leak in patients with secondary spontaneous pneumothorax. A randomized controlled trial , Monaldi Archives for Chest Disease: Vol. 92 No. 4 (2022)
<< < 41 42 43 44 45 46 47 48 49 50 > >>
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/monaldi.2022.2440
https://doi.org/10.4081/monaldi.2022.2440





